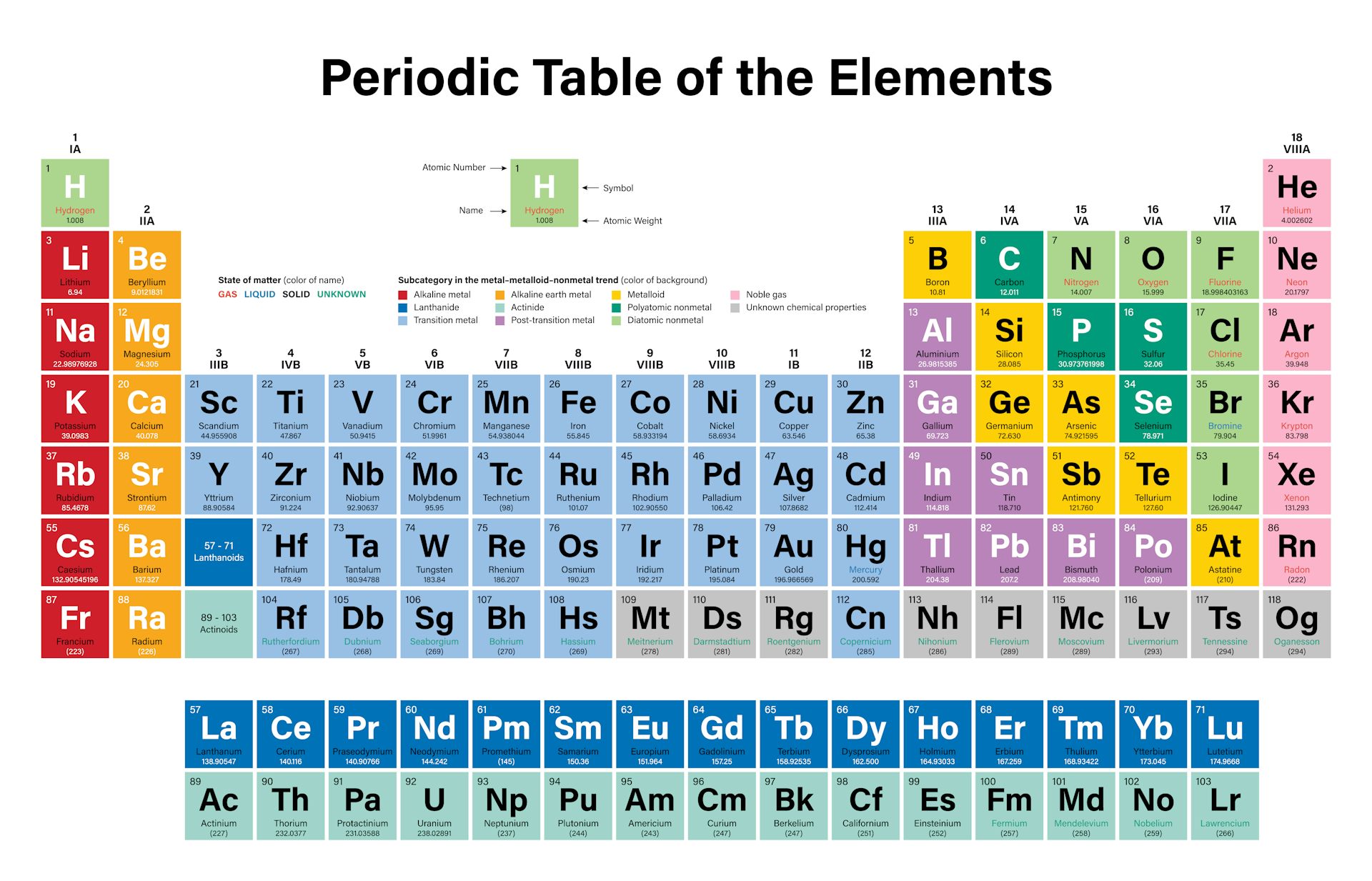
It does not take much to draw splendid colours out of lead. But alchemy also offered lead a chance to shake off its grey and graceless image. The alchemists believed that lead slowly matured into other metals in the ground. In talk of base metals, which alchemy tried to turn to silver and gold, there was none so base as lead. To alchemists, lead was the lowliest of metals - in a sense, it was where all metals started. Political stability of top reserve holderĪ percentile rank for the political stability of the country with the largest reserves, derived from World Bank governance indicators. The higher the value, the larger risk there is to supply.Ī percentile rank for the political stability of the top producing country, derived from World Bank governance indicators. The percentage of the world reserves located in the country with the largest reserves. The higher the value, the larger risk there is to supply. The percentage of an element produced in the top producing country. Low = substitution is possible with little or no economic and/or performance impact Medium = substitution is possible but there may be an economic and/or performance impact High = substitution not possible or very difficult. The availability of suitable substitutes for a given commodity. A higher recycling rate may reduce risk to supply. The percentage of a commodity which is recycled. The number of atoms of the element per 1 million atoms of the Earth’s crust. This is calculated by combining the scores for crustal abundance, reserve distribution, production concentration, substitutability, recycling rate and political stability scores. The Chemical Abstracts Service registry number is a unique identifier of a particular chemical, designed to prevent confusion arising from different languages and naming systems.ĭata for this section been provided by the British Geological Survey.Īn integrated supply risk index from 1 (very low risk) to 10 (very high risk). Where more than one isotope exists, the value given is the abundance weighted average.Ītoms of the same element with different numbers of neutrons. This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). 
The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. (In the modern periodic table, a group or family corresponds to one vertical column.A vertical column in the periodic table. 
The periodic table allows chemists a shortcut by arranging typical elements according to their properties and putting the others into groups or families with similar chemical characteristics. Were it not for the simplification provided by this chart, students of chemistry would need to learn the properties of all 118 known elements. The term “periodic” is based on the discovery that elements show patterns in their chemical properties at certain regular intervals. Mendeleev left spaces for elements he expected to be discovered, and today’s periodic table contains 118 elements, starting with hydrogen and ending with oganesson, a chemical element first synthesized in 2002 at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia, by a team of Russian and American scientists. Its story is over 200 years old, and throughout its history, it has been a subject for debate, dispute and alteration.Īttempts to classify elements and group them in ways that explained their behavior date back to the 1700s, but the first actual periodic table is generally credited to Dmitri Ivanovich Mendeleev, a Russian chemist who in 1869 arranged 63 known elements according to their increasing atomic weight. Go into any scientist’s office or lecture hall anywhere in the world and you are likely to see one. There is no more enduring reflection of science than the Periodic Table of Chemical Elements, which sheds light not only on the essence of chemistry but physics and biology as well.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
